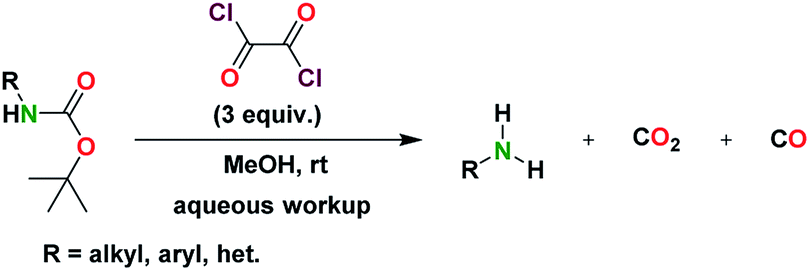
The inherent orthogonality of Fmoc and Boc chemistry allows facile synthesis of branched and cyclized peptides on the resin as well. However, for an activated amine such as a pyrrole or indole, the Boc group can be cleaved A t-butyl carbamate (Boc) group is a common protect- under basic conditions. This protocol thus allows practicing Boc chemistry in a greener manner, and on-demand switching from Fmoc to Boc chemistry and vice-versa while synthesizing one oligopeptide on the same resin. Volumetric measurements of the swelling of the resin in the different deprotection solvents were carried out. Most importantly, this protocol can be used for the solid-phase peptide synthesis (SPPS) using Boc-protected amino acids on the acid-sensitive Rink amide resin, which is usually used for Fmoc chemistry. 3 the selective cleavage of Boc group from N-Boc imidazoles and indoles is achieved with Cs 2 CO 3, imidazole at 70 C 4 and N-Boc-2-lithiated pyrroles are deprotected with NaOMe/MeOH/THF.5 Furthermore, deprotection of N-Boc-indoles with TBAF in refluxing THF has been reported, 6 while removal of Boc protecting group from the aromatic ring of. The major impurity obtained during synthesis when 100 TFA was used for Boc removal corresponded to the omission of the second amino-acid residue added. The solvent was removed in vacuo and the resultant yellow solid was dissolved in MeOH (1 mL). Green Chemistry Overview: Atom Efficiency (By-Products Mwt < 200 g/mole): CO 2 (44.0 g/mole) + isobutylene (56.0 g/mol) 100 g/mole. Boc Protection Ex (HCl) Boc Deprotection (HCl) Examples: Example 1 The SM (80 mg) was dissolved in dioxane (5 mL) and treated with 4M HCl in dioxane (2 mL). A library of di- to tetrapeptides is synthesized with moderate to good yield from standard and nonstandard Boc-protected amino acids in solution. Hot, super heated or near critical water are good solvents for thermal BOC deprotection, but may not readily available in standard chemical processing labs/plants, requiring pressurized reactors or flow chemistry. The deprotected amino group undergoes C−N bond formation with the next Boc-protected amino acid in the presence of a suitable coupling reagent and base. Deprotection is generally achieved under acidic conditions, as extensively described in Greene’s Protective Groups in Organic Synthesis. The general procedure for dM-Dmoc protection of aliphatic amines (i.e., the procedure for the synthesis of 5ad) was used to covert 6 to 7. Don't run boc deprotections in closed systems. The CO2 gas that forms during the reaction should be allowed to escape. 
Boc protecting group is removed from the N-terminal amino acid of the amino acid and the peptide using environment-friendly cost-effective Lewis acid, FeCl 3. Selective deprotection of dM-Dmoc protected amine in the presence of a Boc protected amine: Compound 6 was prepared following a reported procedure. Key Points: The tert -butyl cation will either be quenched by a suitable trapping agent, deprotonate to form isobutylene (gas), or polymerize to form isobutylene oligomers. A mild, practical, and straight forward method for Boc deprotection and its use in peptide synthesis both in solution and on solid support is presented.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
